Chemical Environmental and Molecular Toxicology Lab
The ChEMTox laboratory is part of the School of Science and Technology at the Cupey Campus of Universidad Ana G. Méndez (UAGM). Our facility is located in the Jose F. Mendez Building, Room 125 (First floor). The ChEMTox laboratory supports graduate research and provides valuable research experience for selected undergraduate science students. Our students participate on research projects related to anti-cancer drug development as well as projects in the field of environmental agents with toxicological impact. We implement in-vitro molecular toxicology techniques applying human cancer cells in cultures. We have also developed a new line of research with Zebrafish as an animal model for biomedical research.
In addition to the ongoing research projects, the laboratory offers analytical and technical support to scientists at UAGM, and other academic institutions as an In-vitro biotesting Core laboratory, supported in part by the PR-INBRE project financed by the National Institute of Health. For more information about the In-vitro biotesting Core see attached promo flyer and visit the PR-INBRE site at http://inbre.hpcf.upr.edu/.
Contact Us
- Beatriz Zayas, PhD, MS
Lab Director
787-766-1717 ext. 6462 or 6460
bzayas@uagm.edu
- Karoline Rios, BS, MSEM
Research Associate
787-766-1717 ext. 6462
karios@uagm.edu
By Mail
- ChEMTOX Laboratory
Universidad Ana G Méndez (UAGM)
P.O. Box 21150
San Juan, Puerto Rico
00928-1150
Message from Director

Welcome to the Chemical, Environmental and Molecular Toxicology Laboratory (ChEMTox Lab). We are a very active research laboratory that implement toxicology related research as well as a training place for graduate and undergraduate student interested on gaining research experience that can be applied to the biomedical or environmental toxicology fields.
The ChEMTox facilities at Universidad Ana G. Méndez, Recinto de Cupey was inaugurated on December 2005 and has been developed thanks to the financial support from the National Institute of Environmental Health Sciences (NIEHS), the National Institute of Health (NIH) and Universidad Metropolitana now Universidad Ana G. Méndez, in Cupey. The development of the laboratory represents a key step in the transformation of Universidad Ana G. Méndez, from a primary teaching institution to a teaching-research oriented institution.
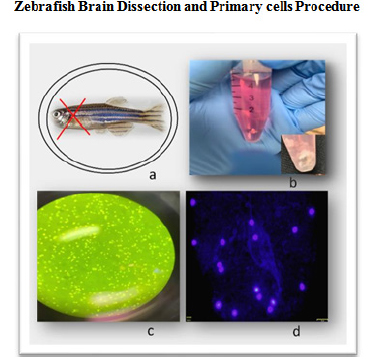
The ChEMTox hosts research projects in drug development as well as environmental toxicology. We apply molecular toxicology methods such as in vitro human cell culture assays. More recently we have implemented a novel zebrafish primary cell culture system by dissection of zebrafish brain tissue. It is important to indicate that all our projects comply with the UAGM research regulatory offices.
Drug Development Research
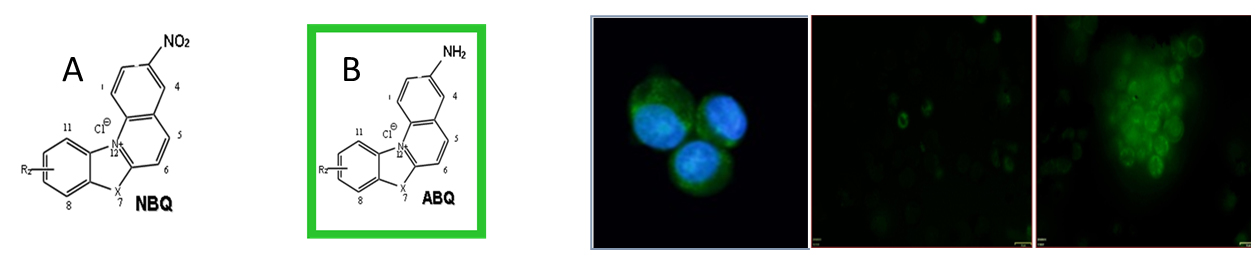
In terms of drug development our group focuses on characterizing the therapeutic potential of novel compounds designed by synthetic or medicinal chemist such as Dr. Osvaldo Cox and other medicinal chemists in Puerto Rico or abroad. Many of our NIH funded project target on drug development that can be applied to cancer treatment or detection. With that purpose we test determining the anti-cancer potential of novel compounds or natural products. A key aspect of such determination is identifying the capacity of these compounds to inhibit the growth of cancer cells in vitro. Also, the interaction of such potential anti-cancer agents with cellular organelles such as mitochondria and DNA. In this context, human tumor and normal cells are treated in culture and their cytotoxicity and cell death mechanism (apoptosis and autophagy) are determined.
The developments of diagnostics markers are also explored at our laboratory as part of the drug development component. Findings in this area have generated multiple scientific publications and four patents for novel fluorescent markers. Currently these patents can be licensed by companies interested in cancer markers.
Environmental Toxicology Research
Understanding the negative effect of environmental agents on human health and other species is a key component of our research. Among many environmental contaminants with human health impact phthalates, polycyclic aromatic hydrocarbons and metals are studied in our laboratory. The presence of phthalates as plasticizer on pharmaceutical products as well as in aquatic environments requires serious attention. Phthalates are compounds widely used in the manufacturing industry and has been associate with multiple adverse health effects to human. Among the family of phthalates many well used for pharmaceutical products. Limited information however has been generated on the effect of DBP on the nervous system. The objective of our ongoing research projects is to determine their neurotoxicity potential applying the zebrafish model as an early stage- screening tool.